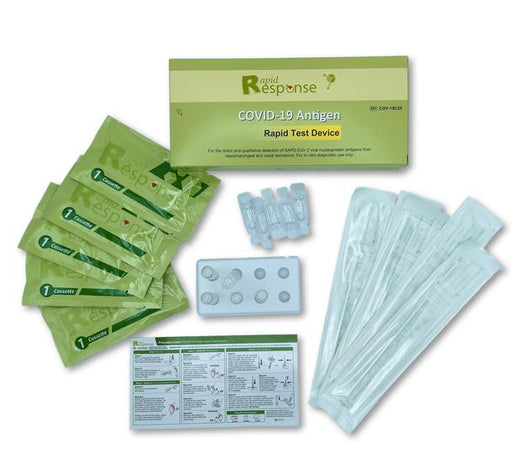
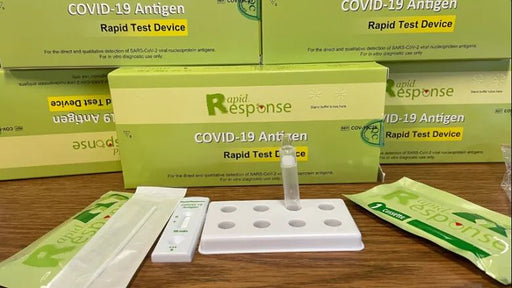
About Rapid Testing Kits & Rapid Antigen Tests
Rapid antigen tests require no special equipment and can be safely used at home to determine a positive or negative result in only a few minutes. Rapid testing works by identifying a protein from the virus that causes COVID-19, however, rapid testing is not as sensitive as a polymerase chain reaction (PCR) test.
PCR tests are highly accurate and can only be analyzed by a healthcare professional in a laboratory setting. When a rapid test indicates a positive result, PCR testing is often used for confirmatory testing.
When Should Rapid Testing be Used?
If You Develop COVID-19 Symptoms
If you develop COVID-19 symptoms, such as fever, dry cough or others, you should take a rapid test as quickly as possible. If you test positive, regardless of your vaccination status, you should self isolate and contact anyone you have recently been in contact with so that they can also take a test as soon as possible.
Although health guidelines vary from province to province and are quickly changing, one negative rapid test has not been considered adequate to leave isolation. It is recommended to have two negative rapid test results (a second rapid test taken 48 hours later), before leaving isolation.
Following Exposure to Someone Who Has Tested Positive for COVID-19
If you were recently in close contact with someone with COVID-19, you should take a rapid test, even if you don’t have symptoms.
To Increase Protection for People at Risk of Severe Disease
If you have friends or loved ones who are at risk of severe disease, it’s recommended to take a rapid test before visiting them, even if you have no symptoms. Likewise, for anyone with a health condition that increases their risk of severe illness, regularly taking rapid tests increases the chances of early detection and treatment, which can help avoid the worst consequences of COVID-19.